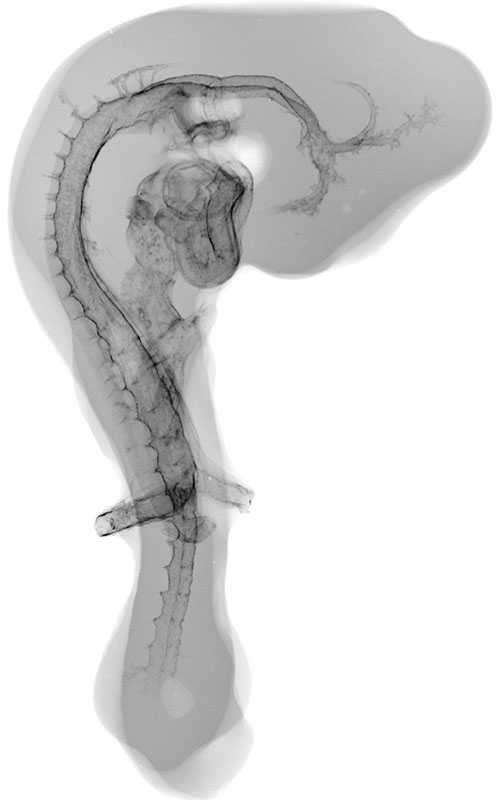
Chick Embryo Microangiography
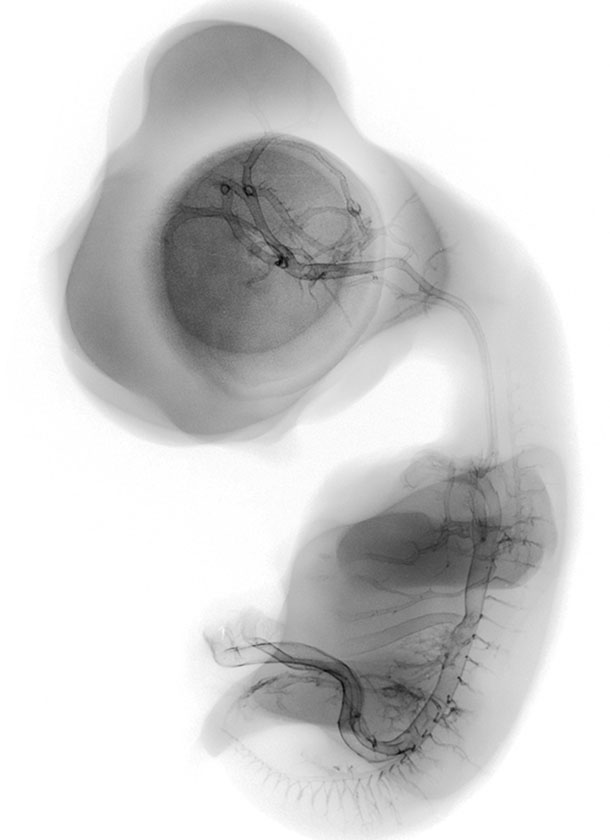

Chick Embryo Microangiography

Methods
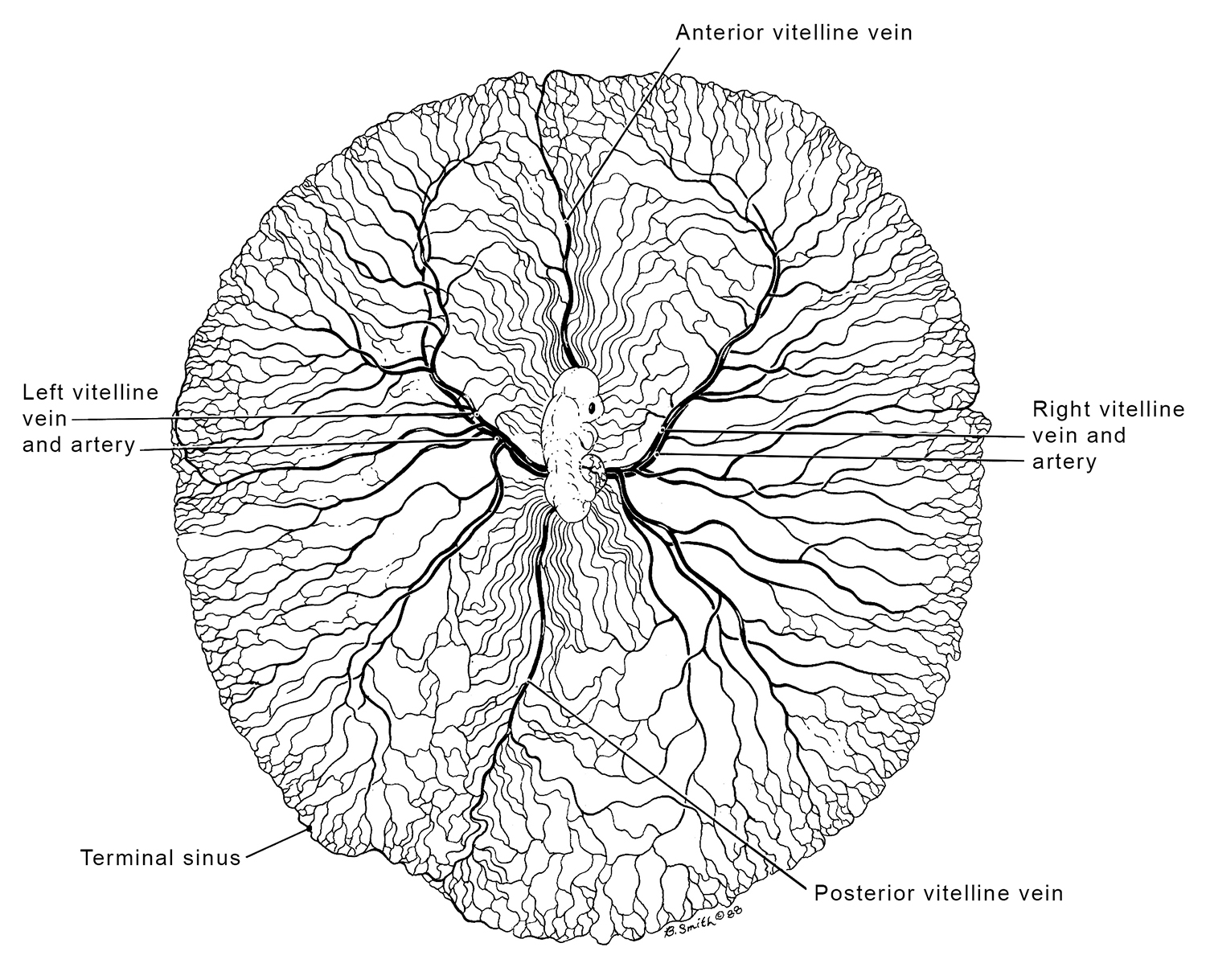
Vitelline Vessels of Stage 24 Chick Embryo
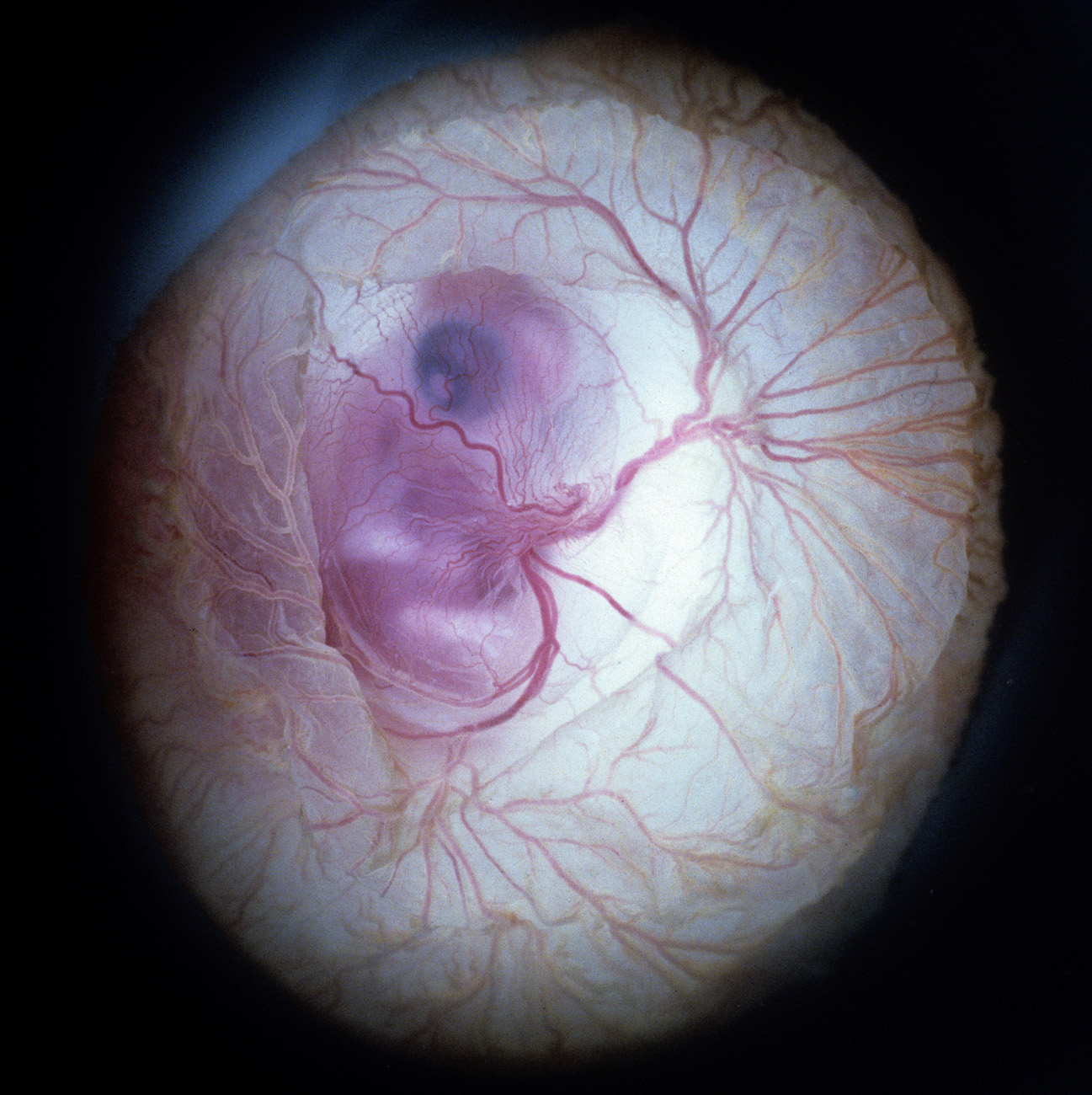
Optical Photos of Stage 30 Chick Embryo
Through Windowed Egg Shell (backlit)
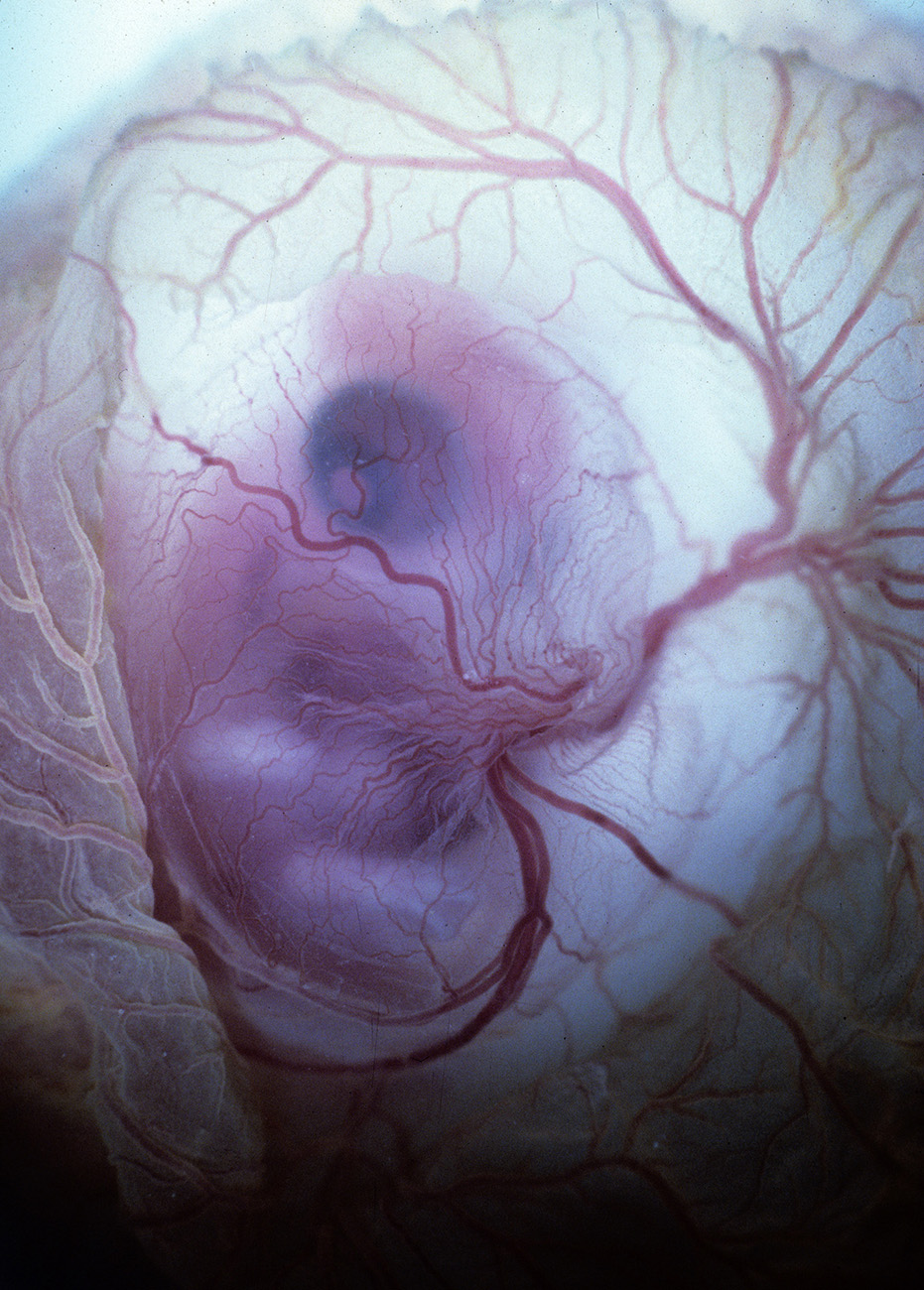
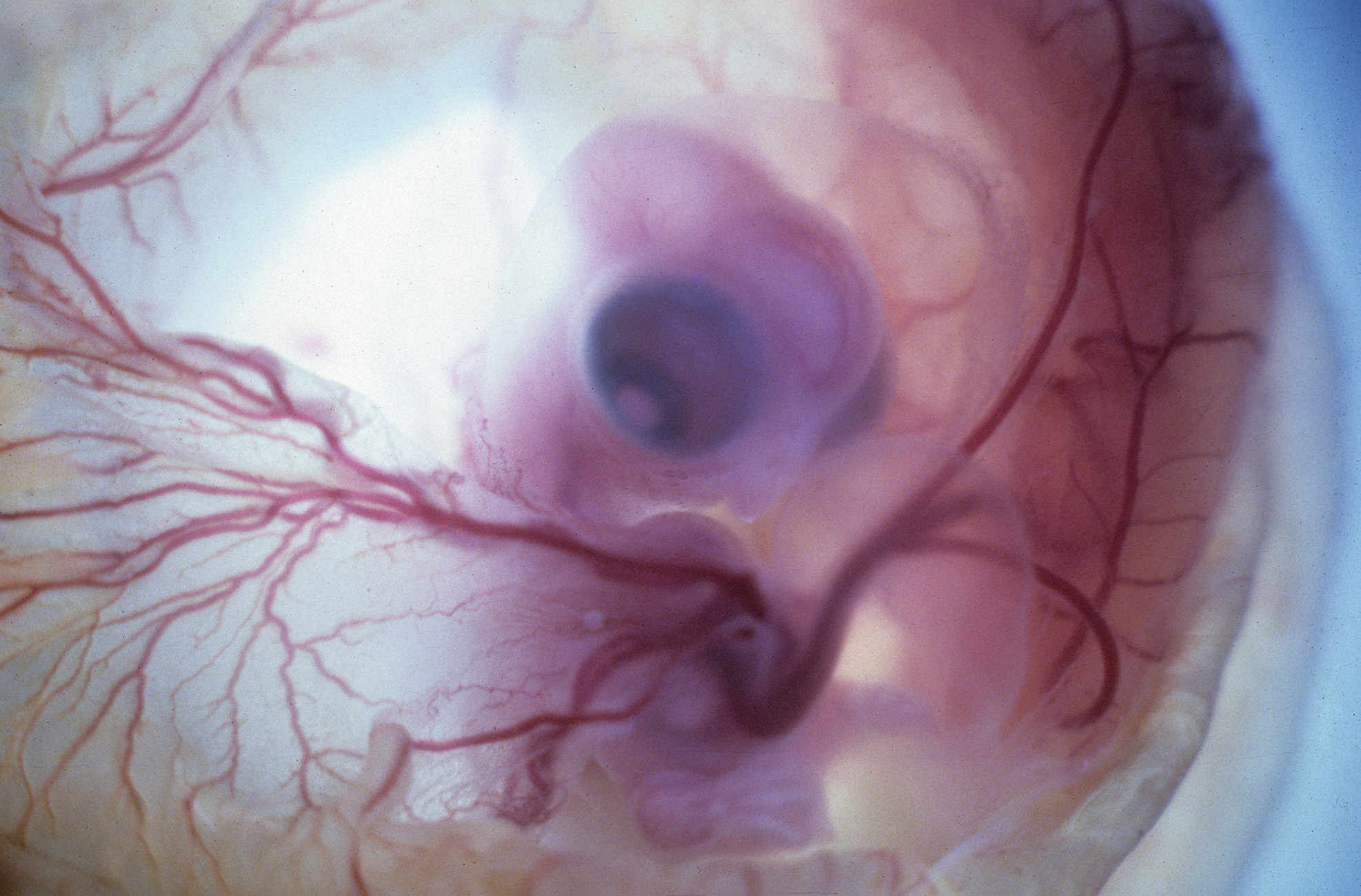
Stereo microangiography of the chick embryos was performed by adapting the methodology developed and reported for mouse microangiography by Eric L. Effmann*.
Fertilized Arbor Acre eggs were maintained in an incubator at 37.5 ºC and 70% humidity with twice daily rotation until the time of vascular perfusion. Windows in the shell were created directly over the embryos for perfusion. Perfusion was performed through a four-channel Gilson peristaltic pump and finely drawn glass micropipette (25-75 micrometer tip diameter) at flow rates of approximately 2-3 microliters per minute. The right vitelline vein was first perfused with 2% glutaraldehyde and 1%formaldehyde in 300 mOsm phosphate buffer supplemented with 0.015% Brilliant Blue dye to visually track perfusion progress. The fixative was followed by a transient perfusion of 2% polyvinylpyrrolidone (PVP) with 0.013% Brilliant Blue and 0.013% Tartrazine, and then by perfusion with 5% silver nitrate. A small tear in the right vitelline vein was created to receive the perfusion micropipette and an additional small tear in the left or right vitelline artery served to vent fluid as the perfusion fluids were introduced. The clear silver nitrate solution reacts quickly with the endothelial cell surface to form a white precipitate in filled vessels signaling the extend of the perfusion. The vitelline vessels were ligated and the embryos immersion fixed in the perfusion fixative (minus the blue dye) immediately following the silver perfusion. The injected silver nitrate reduced to metallic silver for approximately 3 days prior to radiographic imaging.
Microradiography was performed by placing the wet embryos on Kodak High-resolution Plates (type 1-A) in a Hewlett-Packard Faxitron 805 unit operated at 60kVp. The embryo/plate combination was covered with a thin plastic film, and edges were sealed to prevent dehydration. To obtain a second exposure for stereo viewing, the specimens were placed on new film and moved laterally 1.2 cm relative to the x-ray beam. The plates were photographically recorded with an Olympus photo-macroscope.
* Effmann EL, Whitman S, Pexieder T. Stereo microangiography in embryologic and teratologic investigation. Teratology. 1986 Aug;34(1):103-12.
Also see:
Smith BR, Effmann EL, Johnson GA. MR microscopy of chick embryo vasculature. J Magn Reson Imaging. 1992 Mar-Apr;2(2):237-40.